- Language:English
- English


CasNo: 1462-96-0
Molecular Formula: C7H14O
Appearance: Light yellow liquid
|
General Description |
1-Ethylcyclopentanol is a chemical compound with the molecular formula C8H16O. It is a colorless liquid with a minty and floral odor, and it is commonly used as a flavoring agent and fragrance in various consumer products. 1-Ethylcyclopentanol is mainly used in the production of perfumes, soaps, and cosmetics due to its pleasant aroma. It is also used as a solvent in the manufacturing of plastics, resins, and pharmaceuticals. Additionally, it can be used as an intermediate in the synthesis of other organic compounds. While 1-Ethylcyclopentanol is generally considered to have low toxicity, prolonged exposure to high concentrations may cause irritation to the skin, eyes, and respiratory system. |
InChI:InChI=1/C7H14O/c1-2-6-4-3-5-7(6)8/h6-8H,2-5H2,1H3
The present invention relates to the use...
The invention relates to the field of op...
Methods of synthesizing compounds using ...
The expanding “toolbox” of biocatalysts ...
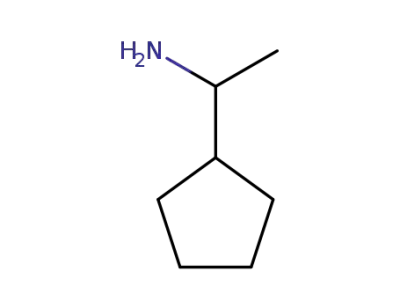
1-cyclopentylethan-1-amine

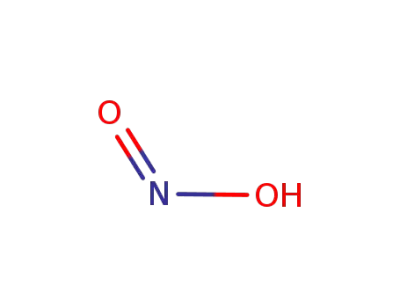
cis-nitrous acid

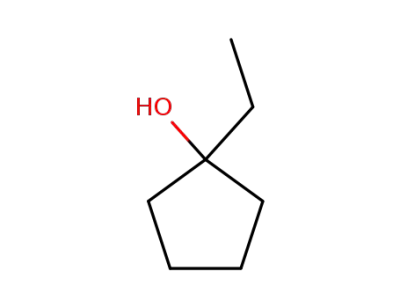
1-ethylcyclopentanol

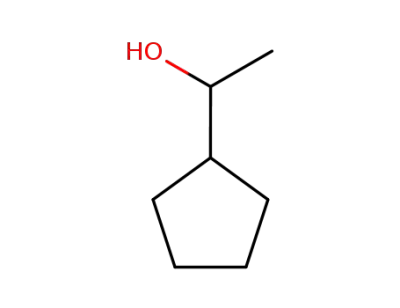
1-cyclopentylethan-1-ol

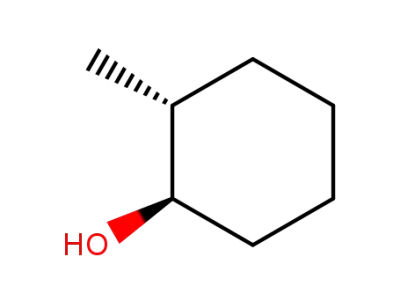
(+/-)-trans-2-methylcyclohexanol
| Conditions | Yield |
|---|---|
|
|
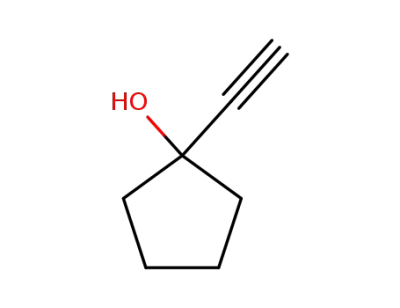
1-ethynylcyclopentanol


1-ethylcyclopentanol
| Conditions | Yield |
|---|---|
|
With
magnesium;
palladium on activated charcoal;
In
methanol;
Ambient temperature;
|
90% |
|
With
5%-palladium/activated carbon; hydrogen;
In
methanol;
at 80 ℃;
under 22502.3 Torr;
Autoclave;
|
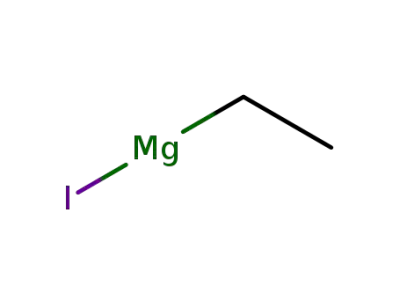
ethylmagnesium iodide

cyclopentanone
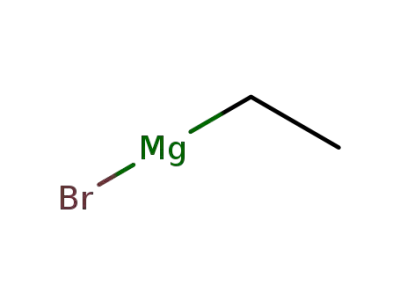
ethylmagnesium bromide
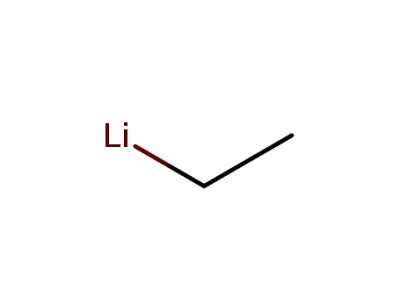
ethyllithium
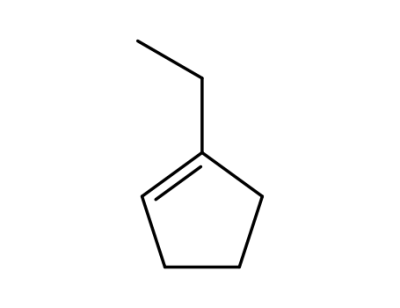
1-ethylcyclopentene
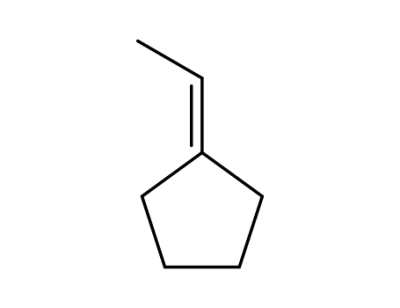
ethylidenecyclopentane
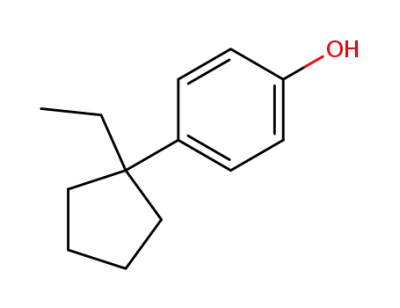
4-(1-ethyl-cyclopentyl)-phenol
1-Ethyl-1-acetamido-cyclopentan
CAS:138071-82-6
CAS:57280-22-5
CAS:22767-49-3
CAS:2832-45-3