- Language:English
- English


CasNo: 1009-11-6
Molecular Formula: C10H12O2
Appearance: white crystal
InChI:InChI=1/C10H12O2/c1-2-3-10(12)8-4-6-9(11)7-5-8/h4-7,11H,2-3H2,1H3
We report the preliminary results of the...
We report the initial results of our stu...
We present herein a novel strategy for t...
Supramolecular catalysts emulate the mec...
A single-step synthesis of 4-hydroxy-fun...
Direct oxidative coupling of phenols and...
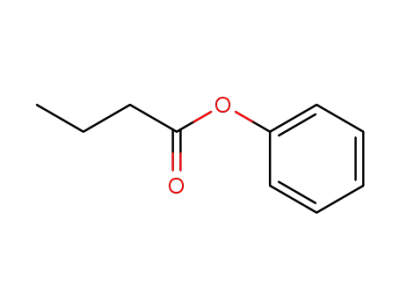
phenyl butanoate

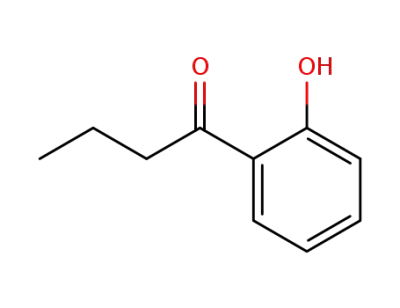
2-hydroxybutyrophenone

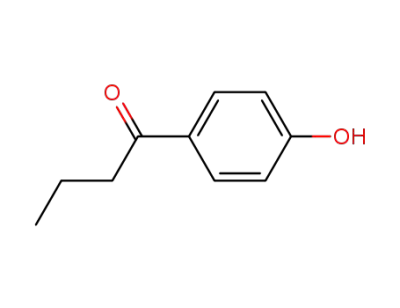
1-(4-hydroxyphenyl)-butan-1-one
| Conditions | Yield |
|---|---|
|
With
aluminium trichloride;
In
nitrobenzene;
|
66% |
|
With
methanesulfonic acid; Methanesulfonic anhydride;
at 65 ℃;
regioselective reaction;
Inert atmosphere;
|
61% |
|
With
aluminum (III) chloride;
In
chlorobenzene;
for 3.5h;
Reflux;
|
51% 43% |
|
With
aluminium trichloride;
In
carbon disulfide;
|
33.4% |
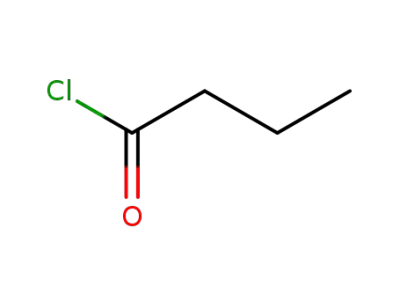
butyryl chloride

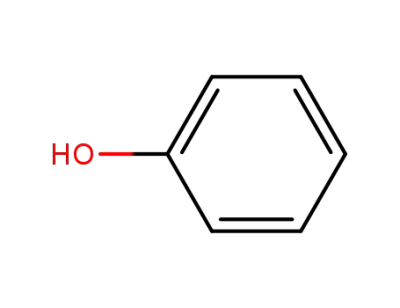
phenol


2-hydroxybutyrophenone


1-(4-hydroxyphenyl)-butan-1-one
| Conditions | Yield |
|---|---|
|
With
aluminium trichloride;
In
dichloromethane;
for 4h;
Heating;
|

butyryl chloride

phenol
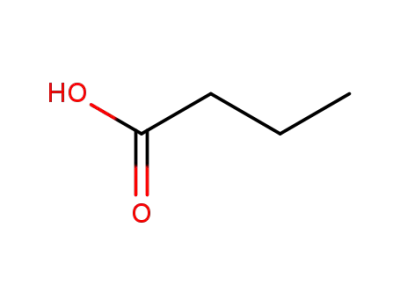
butyric acid

phenyl butanoate
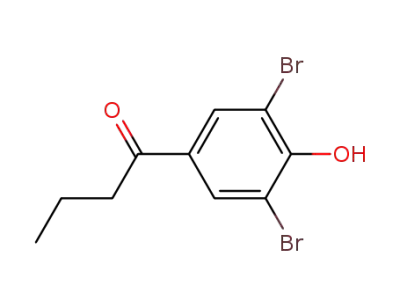
1-(3,5-dibromo-4-hydroxy-phenyl)-butan-1-one
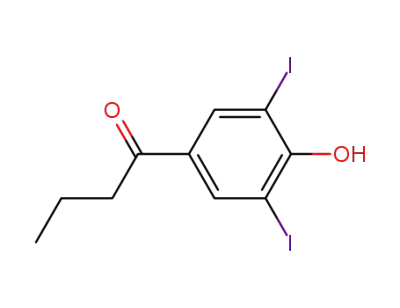
1-(4-hydroxy-3,5-diiodo-phenyl)-butan-1-one
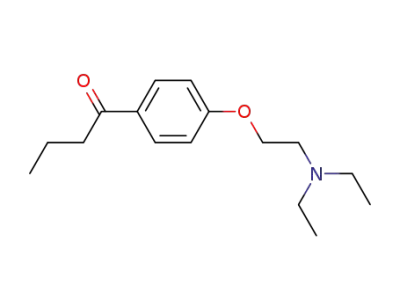
1-[4-(2-diethylamino-ethoxy)-phenyl]-butan-1-one
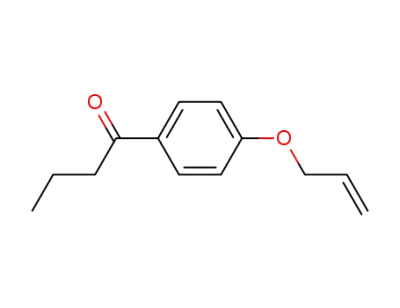
1-(4-allyloxy-phenyl)-butan-1-one
CAS:138071-82-6
CAS:57280-22-5
CAS:56038-13-2
CAS:13361-34-7