- Language:English
- English


CasNo: 501-98-4
Molecular Formula: C9H8O3
Appearance: Off-white to beige or greenish powder
|
Preparation |
Synthesis of p-Coumaric acid: take o-acetylsalicyloyl chloride as raw material, in the presence of magnesium chloride, react with diethyl malonate in acetonitrile solvent, then add triethylamine, and react at 0°C for 1 h to obtain ( 2-acetoxybenzoyl) diethyl malonate product, then heated to 50°C in potassium hydroxide/methanol solution, reacted for 3h, and cyclized to obtain p-Coumaric acid. |
|
Biochem/physiol Actions |
p-Coumaric acid is mainly a plant metabolite which exhibits antioxidant and anti-inflammatory properties. It also shows bactericidal activity by damaging bacterial cell membrane and by interacting with bacterial DNA. In western blot, p-coumaric is also used as an enhancer in chemiluminescent substrate.Hydroxycinnamic acid found in many fruits and vegetables. |
|
Purification Methods |
Crystallise p-coumaric acid from H2O (charcoal). It forms needles from concentrated aqueous solutions as the anhydrous acid, but from hot dilute solutions the monohydrate acid separates on slow cooling. The acid (33g) has been crystallised from 2.5L of H2O (1.5g charcoal) yielding 28.4g of recrystallised acid, m 207o. It is insoluble in *C6H6 or pet ether. The UV in 95% EtOH has max 223 and 286nm ( 14,450 and 19000 M-1cm-1). [UV Wheeler & Covarrubias J Org Chem 28 2015 1963, Corti Helv Chim Acta 32 681 1949, Beilstein 10 IV 1005.] |
|
Definition |
ChEBI: p-coumaric acid is the trans-isomer of 4-coumaric acid. 4-coumaric acid is a coumaric acid in which the hydroxy substituent is located at C-4 of the phenyl ring. It has a role as a plant metabolite. It is a conjugate acid of a 4-coumarate. |
|
Application |
trans-4-Hydroxycinnamic acid is used as a hydroxycinnamic acid with antioxidant properties. p-Coumaric acid is an aromatic phytochemical for proteomics research. It has been used as a component of chemiluminescent substrate for protein detection in western blotting. |
InChI:InChI=1/C9H8O3/c1-6(9(11)12)7-2-4-8(10)5-3-7/h2-5,10H,1H2,(H,11,12)
Photoisomerization of a series of substi...
Cadaba rotundifolia (Forssk.) (family: C...
The MeOH extract from dried whole Sedum ...
The stability of phenolic compounds was ...
![peonidin 3-O-[2-O-(2-O-(trans-feruloyl)-glucosyl)-6-O-(cis-p-coumaroyl)-glucoside]-5-O-[6-O-(malonyl)-glucoside]](/upload/2025/12/c01b7e32-4650-4a29-b381-107b0fc44c10.png)
peonidin 3-O-[2-O-(2-O-(trans-feruloyl)-glucosyl)-6-O-(cis-p-coumaroyl)-glucoside]-5-O-[6-O-(malonyl)-glucoside]

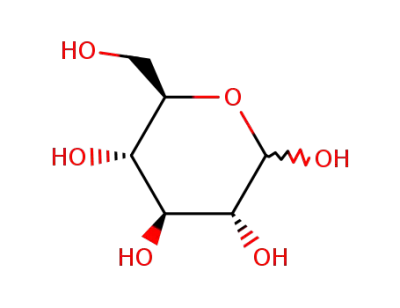
D-Glucose

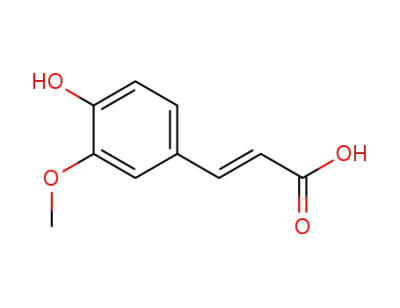
(E)-3-(4-hydroxy-3-methoxyphenyl)acrylic acid

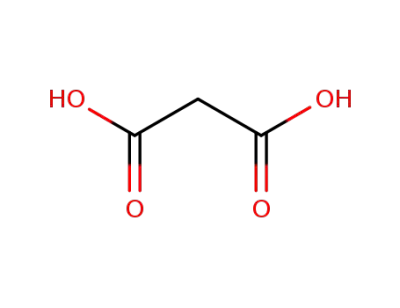
malonic acid

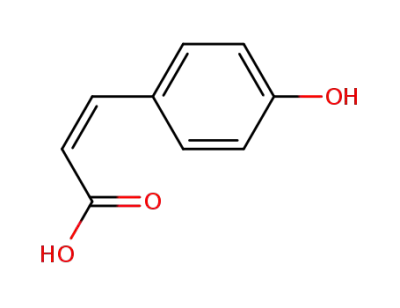
(Z)-p-coumaric acid

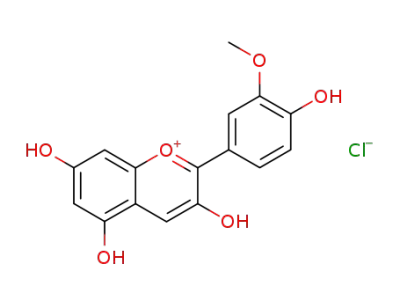
peonidin chloride
| Conditions | Yield |
|---|---|
|
With
hydrogenchloride; water;
at 100 ℃;
for 1h;
|
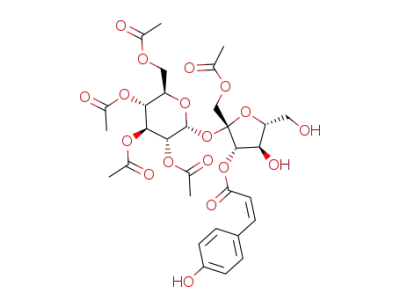
1,2',3',4',6'-penta-O-acetyl-3-O-(Z)-p-coumaroylsucrose

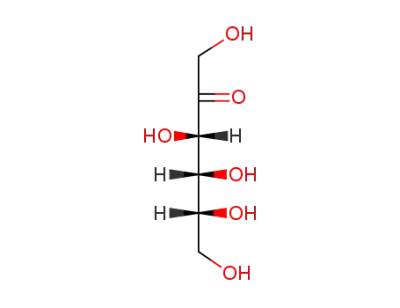
D-Fructose

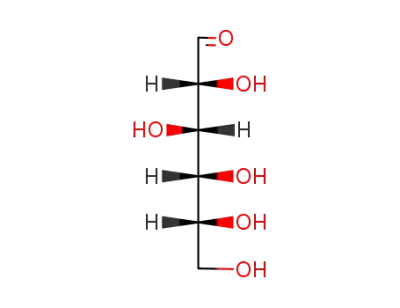
D-glucose


(Z)-p-coumaric acid
| Conditions | Yield |
|---|---|
|
With
sulfuric acid; water;
In
1,4-dioxane;
at 90 ℃;
for 3h;
|
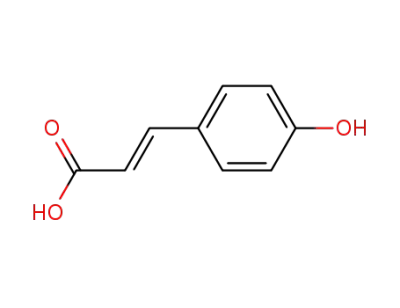
p-Coumaric Acid
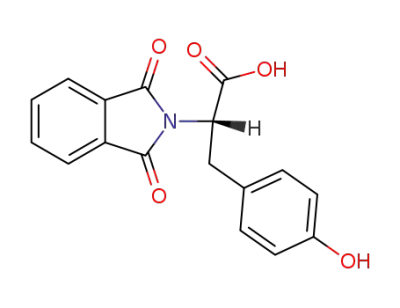
N-phthaloyl-L-tyrosine
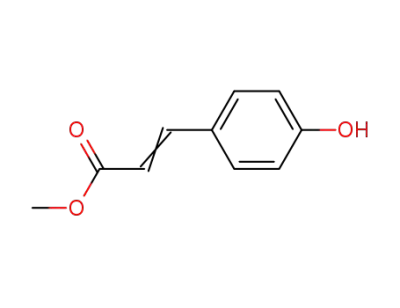
methyl p-hydroxycinnamate
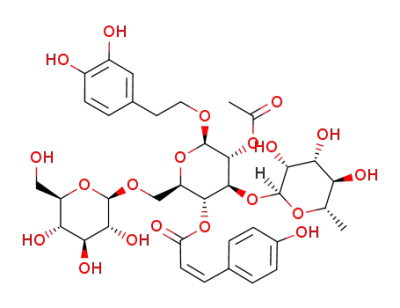
kankanoside H2
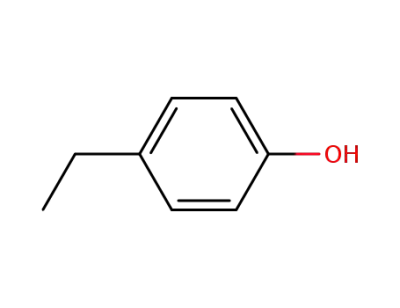
4-Ethylphenol
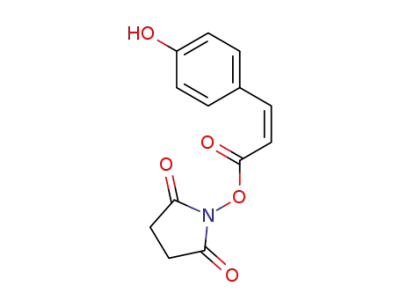
cis-p-coumaroylsuccinimide
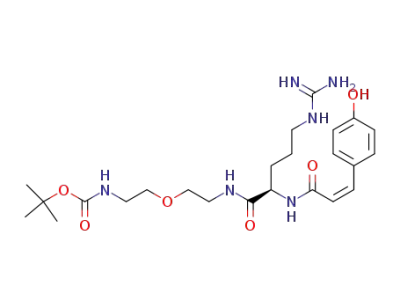
N-(2-(tert-butoxycarbonylamino)ethoxyethyl) Nα-cis-p-coumaroyl-D-arginine amide
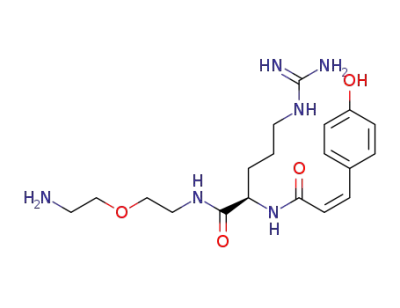
(R)-5-Guanidino-2-[(Z)-3-(4-hydroxy-phenyl)-acryloylamino]-pentanoic acid [2-(2-amino-ethoxy)-ethyl]-amide
CAS:138071-82-6
CAS:57280-22-5
CAS:94686-82-5
CAS:574-12-9